 So ammonia before had the same thing, ammonia has 5 valence electrons so 2, 3, 4, 5, this should be the same I'm sorry they're kind of uneven, they should actually be the same in energy and we have the 5 electrons. So let's look at ammonia as an example, ammonia if you look at nitrogen within ammonia it has these 2 lone pair of electrons. And we also want to say that low in pairs are also going to be hybridized because they're not higher or lower in energy than those bonds either. Because they're hybridized bonds we're going to now call single bonds sigma bonds, this is just the way they overlap, the way that orbitals overlap we're going to call them, denote them sigma bonds. So all single bonds are going to be hybridized. So chlorine is coming here, here, here and here and make 4 of those bonds like you see in the picture.Īlright so what actually would get hybridized? What do we create to actually mix it so that these equal orbitals are necessary? So we know all single bonds are going to be hybridized because a single bond there's not one that's more energetic than the other. And we're going to spread these out just like the rule tells us to and we're going to say okay we have 4 electrons which gives us 4 equal places for chlorine to come in and actually bond with that carbon. 
So we're going to have 4 new orbitals and we're going to call them the 1S and the 3 of them are P so we're going to call it SP3, 1 from S 3 from P. So we're going to hybridize all these orbitals to make 4 equal in energy orbitals. Okay so we want to have 4 equal places where chlorine come in and bond with this carbon. 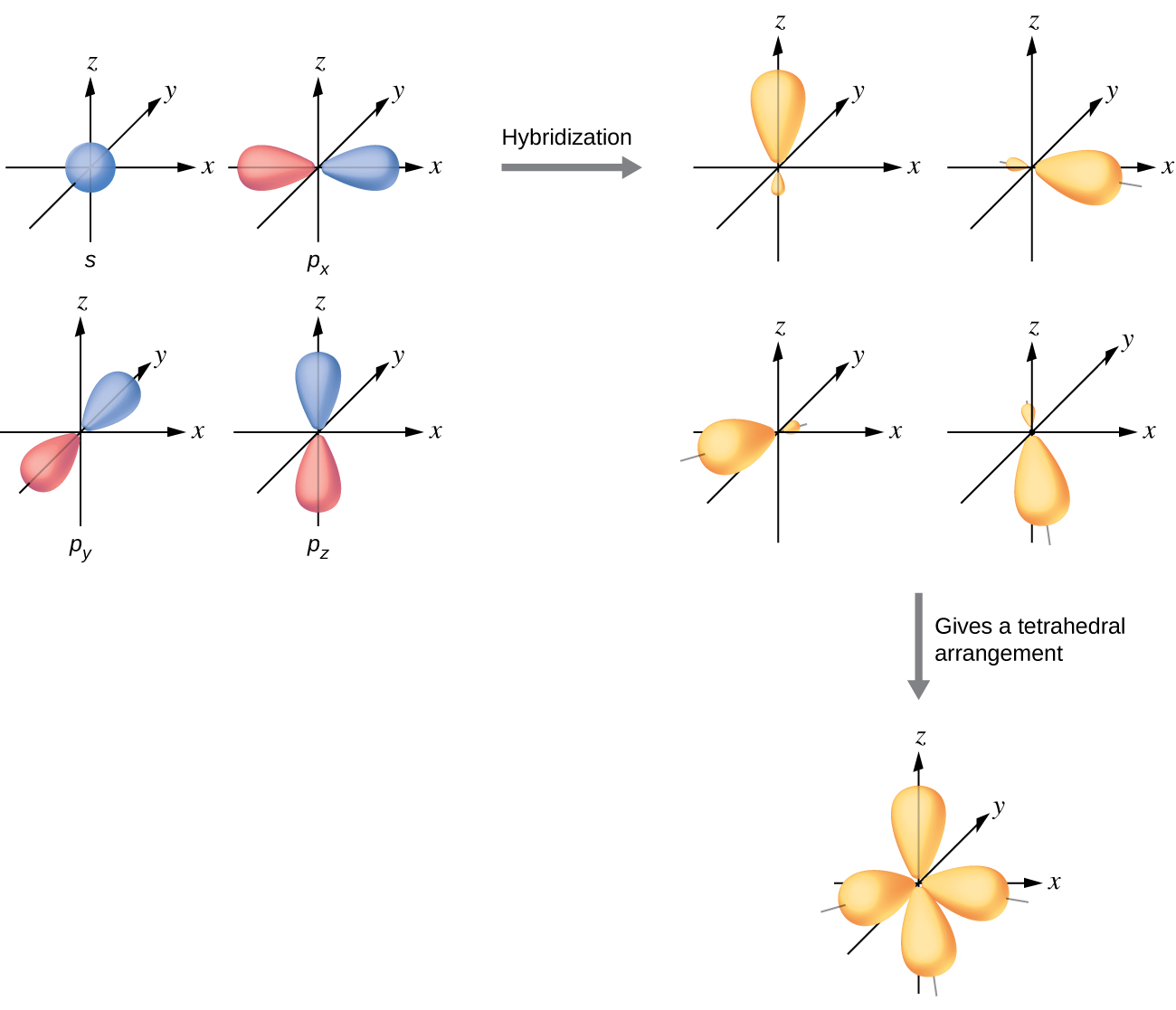
Okay so we know carbon has 4 electrons, we're going to denote them. So how do we make sure that, that's the case? When you look at that orbital, that carbon has in the valence shell we have a 2S orbital and we have 3 equal in energy P orbitals. One is not more energetic than the other, they all have, one is not more special or anything than the other one. So we agree we have to agree that all these bonds, these 4 bonds are equal in energy. So let's start with an example carbon tetrachloride, alright so carbon tetrachloride is a single carbon atom bonded to 4 chlorine atoms. This is a process in which atomic orbitals are mixed to form new identical orbitals this is, are identical in energy.Īlright so let's talk about why we're actually going to do this, why is this even necessary. 
We're going to do the same thing with atomic orbital, we're going to bring 2 different orbitals together to make a completely new orbital we're going to call that process hybridization. Or bikes for example a mountain bike and a road bike they come together to form a new type of bike it's called a hybrid bike. They're 2 ideas of, 2 separate types of cars 1 being electric and one being gas and kind of combine them together to make a new hybrid car it's a completely new different type of a car. 
Alright so we're going to talk about hybridization of atomic orbital and just like you'd think when something is hybridized that you're familiar with let's hybrid cars or hybrid brakes. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
